HSCT vs DMT : What the Global Evidence actually shows — And Why the Answer May Surprise You
Millions of people living with Multiple Sclerosis take their medication faithfully — every day, every injection, every infusion — trusting that modern disease-modifying therapies are the best medicine has to offer. Interferons. Natalizumab. Ocrelizumab. Cladribine. These drugs are sophisticated, expensive, and — let us be honest — only partially effective. They slow things down. They reduce relapses. But they do not stop the disease. They never have.
HSCT — Hematopoietic Stem Cell Transplantation — does something categorically different. It does not slow the immune system’s assault on the nervous system. It ends it. A single course of high-dose chemotherapy eliminates the dysfunctional immune cells responsible for the autoimmune attack, and the patient’s own harvested stem cells are reinfused to rebuild a new, healthy immune system — one that has, in effect, forgotten that it was supposed to destroy myelin. One treatment. Potentially, a lifetime of remission.
But which approach actually wins when you stack the global evidence side by side? And what happens when you look beyond MS — at Scleroderma, CIDP, Lupus, Myasthenia Gravis ? The data is in. It is substantial. And it tells a story that every MS patient deserves to hear.
Two Treatments. – Two Entirely Different Philosophies.
This distinction matters more than most people realise, so let us be precise about it.
DMTs MANAGE. They suppress or modulate the immune system — reducing the frequency and ferocity of attacks on the myelin sheath. The underlying malfunction, however, continues unabated. The immune system is not fixed; it is restrained. Stop the medication and, in the majority of cases, disease activity resurges. Sometimes violently. Patients are, in essence, tethered to their therapy indefinitely, their disease held at arm’s length but never truly defeated.
HSCT RESETS. The procedure ablates the autoreactive lymphocytes — the cells at the root of the immune system’s catastrophic self-attack — using chemotherapy, then reconstitutes the immune system entirely from the patient’s own previously collected stem cells. The regenerated immune system, emerging into a non-inflammatory environment devoid of the “danger signals” that originally triggered autoimmunity, no longer identifies myelin as an enemy. The ambition here is not management. It is elimination.
The MIST Trial: The Most Important Data You Have Probably Never Been Shown
In 2019, the Journal of the American Medical Association published the results of the MIST trial — the Multiple Sclerosis International Stem Cell Transplant randomised clinical trial, led by Dr. Richard K. Burt of Northwestern University Feinberg School of Medicine. It was, and remains, the only properly randomised head-to-head comparison of HSCT against continuing DMT ever conducted.
The design was rigorous. 110 patients with relapsing-remitting MS — all of whom had experienced at least two relapses in the previous year despite being on DMT — were randomised equally. Fifty-five received HSCT using a non-myeloablative cyclophosphamide plus ATG conditioning regimen. Fifty-five continued or escalated to a different, higher-efficacy DMT. The trial ran across four international centres on three continents from 2005 to 2016. These were real patients, with real disease burden, evaluated under real clinical conditions.
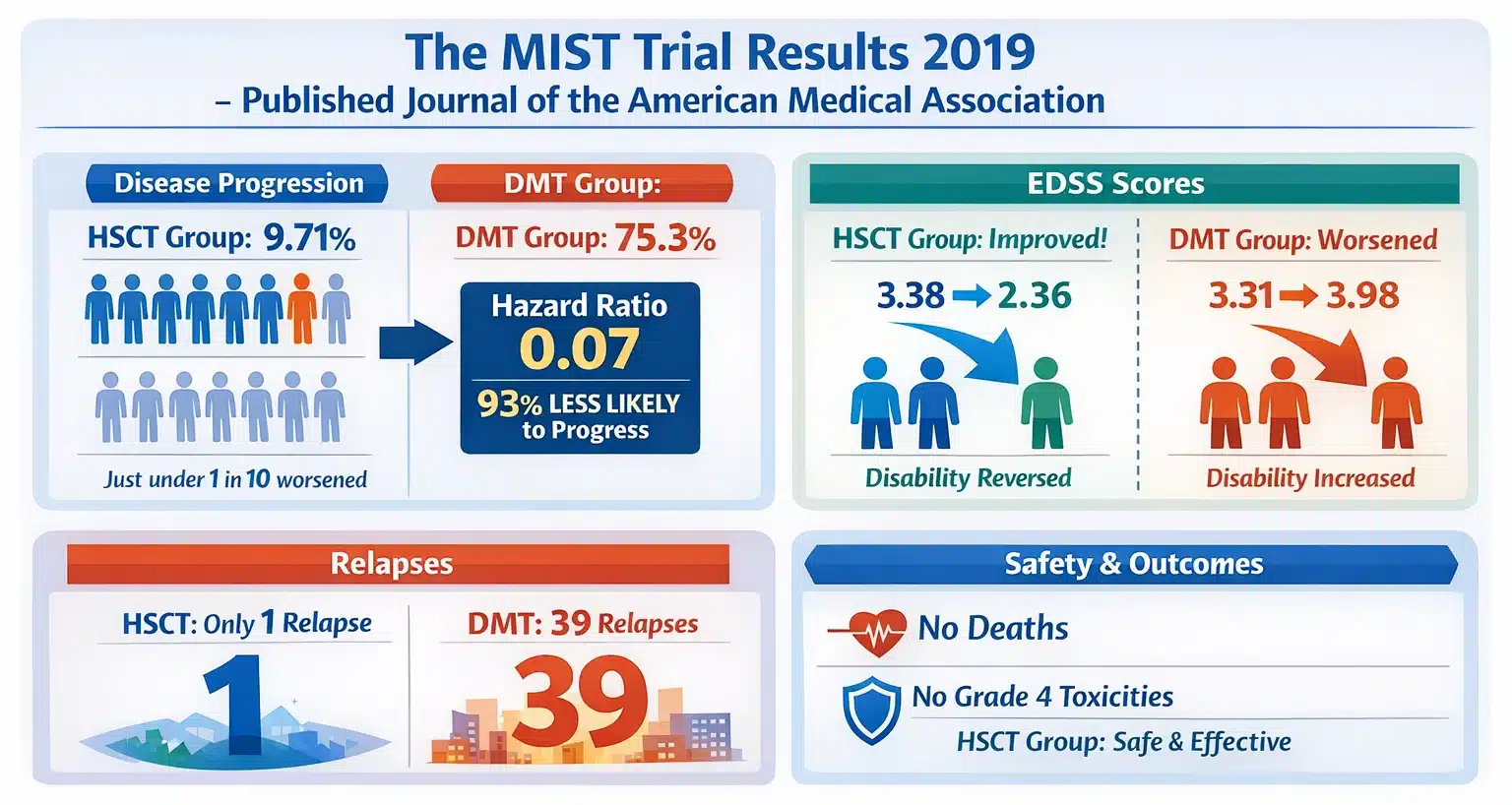
What happened at five years?
- Disease progression — HSCT group: 9.71%. Just under one in ten patients experienced worsening disability over five years.
- Disease progression — DMT group: 75.3%. Three in every four patients worsened. The median time to progression was 24 months — they were deteriorating within two years of the trial beginning.
- The hazard ratio was 0.07. Translation: patients treated with HSCT were 93% less likely to experience disease progression than those who stayed on DMT.
- EDSS scores — HSCT group: Improved. Mean scores dropped from 3.38 to 2.36 in the first year. Disability was not just halted — it was reversing.
- EDSS scores — DMT group: Worsened. Mean scores rose from 3.31 to 3.98 in the same period. The standard-of-care arm was failing while the experimental arm was succeeding.
- Relapses: One. A single relapse across the entire HSCT arm. The DMT arm recorded 39.
- No deaths. No grade 4 non-haematopoietic toxicities in the HSCT group. Safety was not the problem everyone feared.
NEDA-3: The Metric That Exposes Everything
NEDA-3 — No Evidence of Disease Activity, composite measure three — is the most demanding benchmark in MS medicine. To achieve it, a patient must simultaneously have zero relapses, zero new MRI lesions, and zero disability progression. It represents, in short, the goal every MS patient hopes for: a life without disease activity.
Here is what years of published data tells us about how often different therapies achieve it:
| Treatment | NEDA-3 at 2 years | NEDA-3 at 4–5 years |
| Standard DMTs (interferon / glatiramer acetate) | 30–50% | ~18% |
| High-efficacy DMTs (natalizumab, ocrelizumab) | ~55–60% | ~30–35% |
| HSCT — non-myeloablative, leading accredited centres | 70–85% | 65–80% |
Read that table carefully. At four years, the best conventional DMTs maintain NEDA-3 in roughly 18% of patients — fewer than one in five. Even the highest-efficacy modern agents, the ones neurologists now reach for first in aggressive disease, top out at around 30–35% at five years. HSCT, at experienced centres using non-myeloablative protocols, maintains 65–80% at five years.
The 2024 Norwegian long-term study, tracking 29 aggressive RRMS patients for a mean of 70 months after their transplant, found 69% sustained NEDA-3 throughout the entire follow-up window — with 83% remaining entirely relapse-free, and 90% free of disability progression. A 2025 Lithuanian prospective study of 42 highly active RRMS patients over 65 months found 65% maintained NEDA-3, and 35.6% showed genuine EDSS improvement by month three, with that improvement persisting. Critically, those who did experience disease activity after HSCT responded to subsequent DMT. The transplant does not foreclose future options. It simply renders them unnecessary for the great majority.
The Uncomfortable Truth about Timing
One finding in the global literature is particularly inconvenient for the conventional treatment pathway — and arguably the most important discovery any MS patient can encounter. It comes from one of the world’s highest-volume private HSCT centres, which tracked 1,132 patients across all MS subtypes.
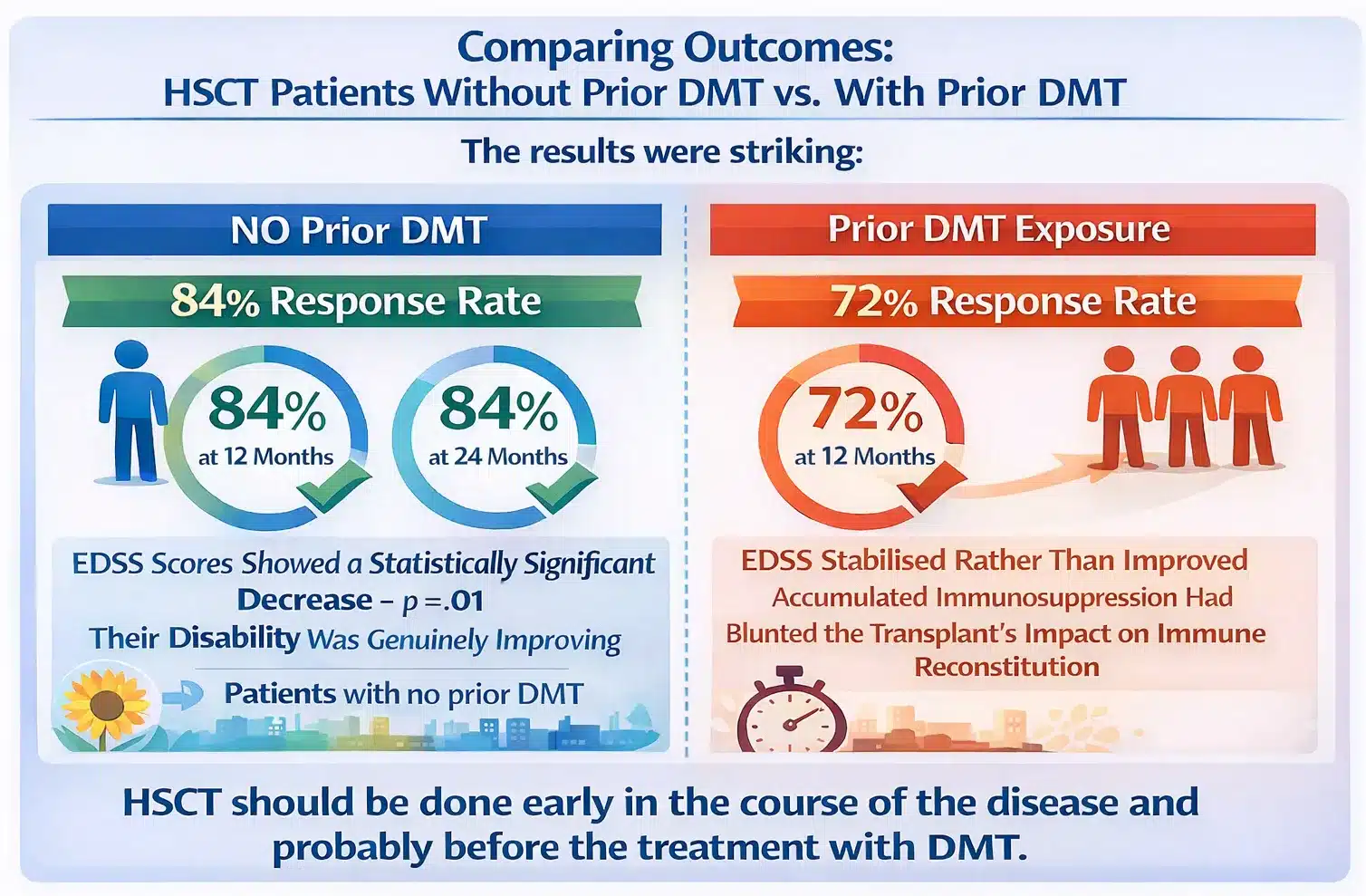
The researchers compared outcomes between patients who had received DMT before HSCT and those who had not. The results were striking :-
- Patients with no prior DMT: 84% response rate (improvement plus stabilisation) at 12 months. 84% at 24 months. EDSS scores showed a statistically significant decrease — p = .01. Their disability was genuinely improving.
- Patients with prior DMT exposure: 72% response at 12 months — lower at the critical early window. EDSS stabilised rather than improved, suggesting accumulated immunosuppression had blunted the transplant’s impact on immune reconstitution.
- “HSCT should be done early in the course of the disease and probably before the treatment with DMT.” — Direct quote from the study authors, published in Multiple Sclerosis and Related Disorders, 2023.
This is a direct challenge to how the medical system currently works. In most countries, the pathway runs: diagnosis, start DMT, fail first DMT, try second DMT, fail second DMT, try third DMT, fail — and only then consider HSCT as a last resort. But the evidence increasingly suggests this sequence may be precisely backwards. Every year spent cycling through ineffective therapies is a year of accumulated neurological damage. A year of scarred myelin. And, it appears, a year of diminishing returns on the transplant that might have worked better earlier.
Beyond MS: What HSCT Achieves Across the Autoimmune Spectrum
Multiple Sclerosis was where HSCT’s autoimmune story began, but it is far from where it ends. Autoimmune diseases have become the fastest-growing indication for HSCT worldwide. The EBMT and EULAR registries have now documented thousands of procedures across an expanding range of conditions. The evidence base is uneven — MS and Scleroderma have the strongest data; rarer conditions rely more on registry analyses and case series — but across the board, the trajectory is encouraging.
Systemic Sclerosis (Scleroderma)
Scleroderma is brutal. It fibroses the skin and internal organs with inexorable efficiency, and conventional therapies have — for four straight decades — failed to shift the mortality curve. Nothing has worked long-term. Until HSCT.
- The ASTIS randomised trial, published in JAMA in 2014, pitted HSCT against 12 monthly cyclophosphamide infusions in 156 patients. At the 10-year mark, HSCT was significantly superior on both event-free survival and overall survival. This was not a marginal finding.
- The SCOT trial, published in the New England Journal of Medicine in 2018, independently confirmed these results, with HSCT producing superior global rank composite scores at 54 months.
- EULAR issued guidelines in 2016 recommending HSCT for rapidly progressive SSc. The American Society for Blood and Marrow Transplantation endorsed it as standard of care in 2018. The EBMT followed in 2022.
- A 7-year Indian cohort study found sustained improvement in skin score, vasculopathy, and gastrointestinal manifestations — with remarkable quality-of-life gains and zero transplant-related complications recorded across follow-up.
Three independent randomised trials. Three converging conclusions. HSCT is now considered standard of care for severe, progressive Scleroderma by every major haematology and rheumatology body in the world.
Chronic Inflammatory Demyelinating Polyneuropathy (CIDP)
Up to 30% of CIDP patients fail to respond adequately to the standard trio of first-line treatments — IVIG, corticosteroids, plasma exchange. For these refractory patients, the conventional medical playbook runs out. HSCT fills that gap.
- A Swedish cohort study of 11 CIDP patients who had failed multiple prior therapies showed meaningful improvement in INCAT and Rankin disability scores within just 2 to 6 months. Motor nerve conduction amplitudes improved by month 4 — objective, measurable neurological recovery.
- A 2023 systematic review and meta-analysis in Therapeutic Advances in Neurological Disorders confirmed durable neurological improvement in refractory CIDP patients treated with HSCT.
HSCT Hospital India treats CIDP patients and welcomes them for evaluation. Click here to read Guide to HSCT Treatment for CIDP
Neuromyelitis Optica and Myasthenia Gravis
Both conditions represent rapidly expanding indications. The EBMT Autoimmune Diseases Working Party has documented favourable responses in NMO. In Myasthenia Gravis, published case series consistently report sustained remission in patients who had exhausted conventional immunosuppressive options. HSCT Hospital India has treated Myasthenia Gravis patients successfully and evaluates NMO cases on an individual basis.
The Big Picture : What 10 Years of Evidence Tells Us

HSCT carries more immediate clinical risk than swallowing a pill. This cannot and should not be minimised. Transplant-related mortality, which in early protocols at inexperienced centres approached 1%, has fallen sharply at high-volume facilities using modern non-myeloablative conditioning. The MIST trial — 55 patients, five years, international centres — recorded no deaths and no grade 4 non-haematopoietic toxicities. Zero.
But the risk comparison with DMTs is more complicated than it first appears. DMTs do not concentrate their risks. They distribute them — across years, across decades, across a lifetime of chronic immunosuppression. Progressive multifocal leukoencephalopathy (PML) with natalizumab. Rare but potentially fatal infections. The slow, grinding accumulation of side-effect burden. And the financial weight — the MIST trial reported an annual DMT cost of approximately $65,000 per patient in 2013 dollars. That cost does not stop. Ever.
DMTs have their place. For newly diagnosed patients with mild, well-controlled disease, they remain a reasonable and often appropriate first intervention. But for those with aggressive, active, relapsing disease — or for those watching their disability creep upward through a succession of medications that were supposed to hold the line — the global evidence makes a clear and compelling case that HSCT is not the last resort. It may, in fact, have been the right answer all along.
- Burt RK et al. Effect of nonmyeloablative HSCT vs continued DMT on disease progression in RRMS: a randomized clinical trial. JAMA. 2019;321(2):165–174.
- Kvistad CE et al. Autologous HSCT for MS: long-term follow-up data from Norway. Multiple Sclerosis Journal. 2024. doi:10.1177/13524585241231665.
- Sanchez-Bonilla D et al. Long-term results of autografting persons with MS are better in those not exposed to prior DMT. Mult Scler Relat Disord. 2023;75:104744.
- Alexander T et al. Autologous HSCT for rheumatic diseases: EBMT best practice recommendations. Bone Marrow Transplantation. 2025. doi:10.1038/s41409-025-02695-y.
- Inglese M, Cohen JA, Sharrack B et al. Ongoing randomized clinical trials on HSCT in MS. Handbook of Clinical Neurology. 2024;202:307–315.
- Sullivan KM et al. Myeloablative autologous stem-cell transplantation for severe scleroderma. N Engl J Med. 2018;378(1):35–47.
- MS Society UK. HSCT research evidence — StarMS, RAM-MS, BEAT-MS trial summaries. mssociety.org.uk. 2024.
- EBMT/EULAR Position Statement. General information for patients considering HSCT for severe autoimmune diseases. Bone Marrow Transplantation. 2019. doi:10.1038/s41409-019-0430-7.
Why Choose HSCT Hospital India for Your Long-Term MS Treatment
The research is clear: outcomes are best at high-volume, accredited centres with experienced transplant teams. Clinical outcomes are strongly linked to experience and facility standards. HSCT Hospital India has been delivering HSCT for MS since 2016 at #1 JCI-USA accredited World Class Hospital In India .
 HSCT Hospital India has been delivering HSCT for MS since 2016 at #1 JCI-USA accredited World Class Hospital In India.
HSCT Hospital India has been delivering HSCT for MS since 2016 at #1 JCI-USA accredited World Class Hospital In India. Treatment is based on the Dr. Richard Burt non-myeloablative protocol – the same protocol that produced the landmark JAMA trial results.
Treatment is based on the Dr. Richard Burt non-myeloablative protocol – the same protocol that produced the landmark JAMA trial results. JCI-USA Accredited, NABH & NABL certified – the highest standards of patient safety and clinical governance.
JCI-USA Accredited, NABH & NABL certified – the highest standards of patient safety and clinical governance. HEPA-filtered private rooms with triple-level air filtration stays, eliminating infection risk during the critical engraftment phase. There are no outside hospital stays.
HEPA-filtered private rooms with triple-level air filtration stays, eliminating infection risk during the critical engraftment phase. There are no outside hospital stays. All-inclusive 30-day in-hospital package at $30,000 USD – the most transparent and affordable pricing of any accredited HSCT centre globally.
All-inclusive 30-day in-hospital package at $30,000 USD – the most transparent and affordable pricing of any accredited HSCT centre globally.




